
Patination for Small Scale Studio Use
13 Minute Read
In recent years there has been an increased interest in patination and metal coloring. This may be seen as a response to the use of nonprecious metals in jewelry. A trend towards objectmaking in North America has produced relatively large surface areas in base metals that invite patination of various kinds.
Richard Hughes and Michael Rowe's monumental work the Colouring Bronzing and Patination of Metals appeared in 1982, providing carefully researched compilation of coloring solutions and application methods superior to anything previously available on the subject. However, many of the solutions and chemicals are unsuitable for the average small studio. Some are toxic and corrosive and others impractical to make because of difficulties in obtaining small amounts of the required chemicals.
This abbreviated paper addresses the need for simple, less toxic solutions for small-scale studio use. Since there are thousands of solutions, pastes and atmospheres that affect metal surfaces, there is often more than one method of achieving the same or similar color on a specific metal. As this paper is intended as an introduction to metal coloring, basic procedures are emphasized.

Patinated surfaces should not be worn in contact with the skin or used in anything connected with food. Many patinas are toxic if ingested and may cause skin irritation in close or constant skin contact. Protective sealers are often used with patinas. They must be used if the object will be in body contact. The irresponsible use of patinations in the fashion industry is likely to lead to a backlash when the first major skin reactions occur in customers. When used in jewelry, patinas should be isolated from all surfaces in skin contact. They must adhere well to the metal and be properly sealed.
Natural patinas formed on copper alloys vary with the environment. Industrial environments tend to produce a copper sulfate base, the ocean a copper chloride base and the mountains a copper carbonate base. Varying conditions produce combinations of these and others. Natural patinas include:
| Black | Copper Sulfide | Cu2•CuS | |
| Bright Blue | Copper Hydroxide | Cu(OH)2 | |
| Green-Blue | Basic Copper Carbonate | CuCO3•Cu(OH)2 | |
| Green-Blue | Basic Copper Sulfate | CuSO4•3Cu(OH)2 | |
| Green | Basic Copper Chloride | CuCl2•3Cu(OH)2 |
These patinas take four to 30 years to develop well, depending on the location. All of them can be produced faster with chemical treatments. The best durability, color control and speed of application are produced using solutions and patinas that rarely occur in nature.

The oldest traditional method of obtaining green patinas on copper alloy surfaces was to expose them to urine, often aged to developed a stronger ammonia content. Works have been buried in manure, coated with pastes containing urine or sealed in urine atmospheres. I have heard of a Korean metalsmith who had over 40 dated jars of aged urine in his workshop, the older ones being more prized. Diet and physical condition would add minerals, salts and chemical compounds to the solution and subtly affect metal surfaces. Burial in alkaline or acidic earths has also been used to create patinas and may be considered a kind of paste application.
Patination agents included in this paper are clear household ammonia, vinegar, table salt and cupric nitrate. Ammonia and vinegar produce good results as fumes. Salt functions as a direct corrosive agent. A combination of salt and ammonia fumes makes for fast results but adhesion is not as good as with the cupric nitrate patina.
In order to organize the materials, we will deal with the process in the order in which the patination proceeds:
- Cleaning
- Application Techniques
- Masking
- Chemical Agents
- Hot Applications
- Color Options
- Protective Coatings
Cleaning
A clean surface is essential. All mineral and organic oils, greases and oxidation must be removed. The process chosen should activate or roughen the metal surface.
There are four common methods for cleaning the surface: chemical, solvent/detergent, mechanical and electrolytic. One or more of these techniques may be required in any given situation.
- The article is fully annealed and pickled in a conventional acid pickling bath. This may not remove all of the oils present. This process would typically be followed by abrasive treatment and/or cleaning with solvent or detergent.
- Solvent/Detergent. Cleaning in strong solvents or a warm mixture of household ammonia and detergent may be required to remove all traces of grease and oils. For safety reasons, ammonia and detergent are preferred.
- The mechanical process removes oxides and oils and roughens the surface. This increases the surface area to provide improve reactivity and adhesion. Pumice, wet or dry abrasive paper and sandblasting are effective mechanical treatments.
- This type of treatment would include both electrocleaning and electrostripping. A simple electrocleaning solution is:Sodium carbonate 50g
Trisodium phosphate 25g
Sodium hydroxide 25g
Water 200ml

The solution is used at 60-70C. and a current density of 1-3A/dm2. The work is treated for 1-3 minutes as a cathode and 5-10 seconds as an anode. Dip in 1:10 sulfuric acid to neutralize the electrocleaning solution and activate the surface (15 sec.). Rinse well in running water.
The clean part may be stored for a period of time in distilled water to inhibit the reformation of oxides.
Application Techniques
The final appearance of the object is influenced by both the chemical agents used an the application technique. The following details some of the typical applications methods.
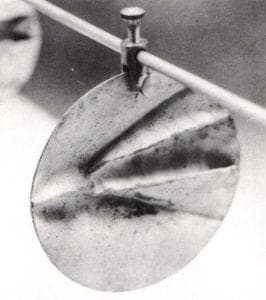
- Cold Fuming. This technique produces an even color except where condensation causes pooling on the surface. Place the object on a platform (to keep it out of the patination agent) in a closed plastic container or bag and pour the agent on the bottom of the container. It is sealed and allowed to react with the metal. Cold fuming works well for ammonia and vinegar.
- Painted Solutions. The solution is patinated on with a swab or brush. A small amount of wetting agent (detergent or alcohol) aids in obtaining even results. The metal dries and the process is repeated until the results are satisfactory. Heat may be used to accelerate the process. Suitable for salt solutions and cupric nitrate patina.
- Sprayed Solutions. This method may be used to obtain an even or speckled effect. Repeated applications and time yield good results. Keep applications light. Spraying works well for salt and cupric nitrate solutions.
- Bound Materials. When a porous material moistened with a patination agent comes in contact with a metal surface, a reaction occurs at the contact points. An object may be wrapped in cloth or string to utilize this effect. Organic materials such as feathers and plants may be bound to the object. If tightly bound, surface activation is prevented and outlines are emphasized. Dry materials bound to the surface and exposed to ammonia or vinegar fumes act as resists. The agents soon permeate the binding medium, and etching as well as patination may take place at the contact points. If brass is left in contact with ammonia or vinegar for long periods of time, it will become fragile and crumble with light pressure. Use other metals for structural elements if using these agents on work.
- If the agent is pooled on the metal it reacts locally. Grease (fingerprints) can emphasize pooling effects. The edges of pools will react differently than the centers.
- The object is immersed in the solution. This method is inappropriate for ammonia or vinegar but may work with salt solutions.
- Moistened Shavings. Damp (not wet) wood shavings or other porous materials work well for differential patination/etching of surfaces. Again, contact points are the focus of action, an example being the polka dots produced by hamster cage cedar shavings. Etched texture is possible on copper alloys with ammonia and vinegar. Possible media include resin-free sawdust and shavings (no oak either), peanut shells, kitty litter, styrofoam balls or chunks, crushed cork, sisal or coarse fibers, pin needles, grass, leaves, straw, etc. Use in a sealed container. The pieces may be shaken to obtain a randomized effect. Rowe and Hughes suggest making a dam of modeling clay to contain the moist media to obtain patterns or masks areas.
Masking
Tape, wax, lacquers and other resists may be used with these agents. Layered metals like mokume, doublée and selectively plated surfaces can be considered as a type of resist. Different reactions occur when the various metals are exposed to the agent. Gold is nonreactive and serves as a good resist. It retains its bright yellow color against the green of the base metal below. "Gold Painting." a gold fusion technique on copper, provides a useful way of developing patterns using gold as a resist.
Chemical Agents
Next we will consider some common agents. Clear household ammonia produces greens and blues on copper alloys, olive greens on bronze. It works best in thin films. This may be provided by treating the object in an ammonia atmosphere with a damp surface. (Without prior dampening it will condense on the metal.) If the metal is wetted with a salt solution rather than water, a bright blue is produced in about two hours. If left overnight, it intensifies but is far less adhesive. Vinegar works in a way similar to ammonia. Moistened shavings work well as an application technique.
Salt may be sprinkled as dry crystals and then left in a humid place (laundry room) to react with the metal. A day or more is required. Solutions of salt produce varied results. Salt patina is characterized by variegated bright green and orange on copper. Salt also works well in combination with other solutions and fumes. An example is the Rowe/Hughes solution #3.129 (cupric nitrate 200g, salt 200g, water 1000ml), which may be applied hot or cold and produces azure and orange surfaces. This produces a loose patina good for non-wearable work, unless well sealed or built-up.

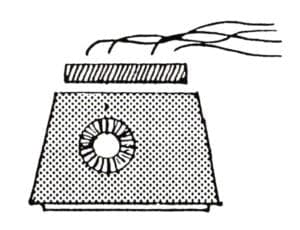
The balance of this paper will deal with the application of cupric nitrate patinas. This general-purpose patina may be applied hot or cold and may be built up to cover surfaces evenly with excellent adhesion. The solution is most easily applied to brass but holds better on copper. It is a surface covering and may be used on silver, nickelsilver, bronze, steel, iron and even gold. Caution: a good fume hood is necessary when working with this chemical. Nitric acid fumes are released (corrosive, irritant) and the possibility of metal-fume poisoning exists. Plans for a small-scale fume hood for patination designed by Theo Jansen and James Evans are available from the Ontario Crafts Council in Canada. To test air-flow speed use a smoking rope to see the draft in action. The smoke should rise at an angle of no more than 10° from the metal as it moves toward the vent. Rubber gloves and cleanliness are essential. This patina should not be used on surfaces next to skin. No eating, drinking or smoking when working with this patina.
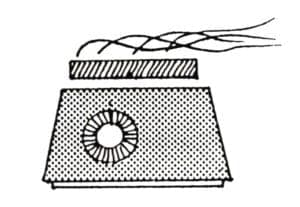
The steps in the application are:
- Prepare metal. Rub well with pumice to remove oxides and roughen surface to aid mechanical adhesion.
- Clean metal to remove grease and oil.
- Place flat work on heated plate. If the work is three-dimensional, place it on clean firebrick or turntable to heat evenly with torch flame.
- Moisten felt pad and brush it on almost dry. The solution should dry instantly. If spraying, a light touch is suggested. Spraying on hot metal is especially dangerous, waste is high and containment of excess spray essential.
- Heat surface until it turns darker, to brown. Expose it to a flame, burning the layer on, turning it black. Too much heat burns off the layer.
- With a stiff brush under running water remove all loose material and repeat until the surface is an even green. The removal of loose material is what leads to good adhesion in the finished surface. To obtain a mottled effect scrub harder or don't clean well before starting. A dip in a 2% hydrogen peroxide solution between applications improves adhesion. To dry use a gentle heat.
Repeated applications and removal of all loose material by scrubbing in water between exposures is recommended for the best adhesive qualities.
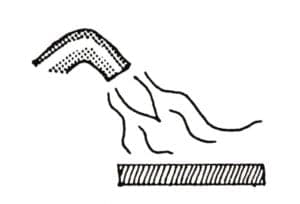
Color Options
A wide range of surface colors are available from the more common agents:
- Yellowish tone: Remove metal from heat before patina turns green.
- Brownish yellow on green: Expose to a light flame. High areas of texture will turn brown.
- Green with brown outlines: Lay out pattern with water or a gel such as KY® or Lubafax.® A flame played over the surface turns exposed areas brownish with a brown outline.
- Dark Brown: Bleach patined on; 30-second exposure. If immersed, a mottled brown and green results.
- Greener: Add a few drops of acetic acid (vinegar)-do not add too much as it may ruin the solution.
- Blue-green to blue: Dip in a weak lye solution.
- Bluish green to turquoise: An easy variation, expose the work to ammonia fumes or paint it on. Heavilty treated areas may lose adhesion. Apply to cool metal. The color reverts to green upon reheating.
- Black: This is a repeated application burnt black each time, reported by Anne Graham.
- White (very pale green): Vinegar painted on; approximately 60 seconds working time.
- White: 1:2 nitric acid solution (fumes!). If left on it removes the patina down to the metal surface.
- Bright metal: May be obtained by scraping through the patina layer (dust hazard; wear a good dust mask). Paint, inks or gold leaf may be laid in the incised areas or lines.
Protective Coatings
Correctly applied, the patina is tough and not liable to damage easily. Sealers are necessary for jewelry use. These would include waxes, thick oils, acrylics, lacquers, etc. Acrylic resins and resins like Envirotex® are good for over-all thick coverings. Samples should be made as a palette to choose from. Oils and waxes tend to darken the color. acrylics to make it resemble green paint. I usually spray the latter from a distance for a subtle effect and matt the surface with a cloth before it dries. Tapes, string, leaves, etc. may be used as masks to obtain patterned or mottled areas.
It is worth noting that a number of metal coloring procedures and patinas build up a layer of material over the surface, effectively hiding it. The only difference between them and the use of suitable paints is recent historical context. There is no reason, save tradition, why paint, sealed colored pencil, plastic resins and other durable surface coverings should not be used. Where esthetic choice demands and a durable medium is available, that medium should be used if appropriate to the intent of the work. Paint, for example, is faster to apply than many patinas and presents far fewer safety problems. Let the demands of the piece decide surface coverings and not tradition. And if you choose to use patinas care and enjoy the process.
Notes
- Further information may be obtained from the full paper published in the Australian journal Lemel in three parts in 1987 or the paper can be ordered from Charles Lewton-Brain, 120 9th, N.W., Calgary, Alberta, T2N 1S9, Canada. For specific colors, patinas and subtle surface effects, refer to The Colouring, Bronzing and Patination of Metals by Richard Hughes and Michael Rose, London: Crafts Council, 1982. This book also includes an exhaustive bibliography of sources dealing with metal coloring and patination.
- Chemishce Farbungen von Kupfer und Kupferlegierungen, Kupfer Institut, Berlin, 1974, p. 36.
- The Colouring, Bronzing and Patination of Metals, op. cit., p. 41.
- Ibid; p. 194.
- Information on ventilation systems compiled by Jansen and Evans for the 1983 "Good as Gold" conference. Contact: The Ontario Crafts Council, 346 Dundas St. West, Toronto, Ontario, M5T 1G5, Canada.
Charles Lewton-Brain is a jeweler currently teaching at the Alberta College of Art.

The All-In-One Jewelry Making Solution At Your Fingertips
When you join the Ganoksin community, you get the tools you need to take your work to the next level.









