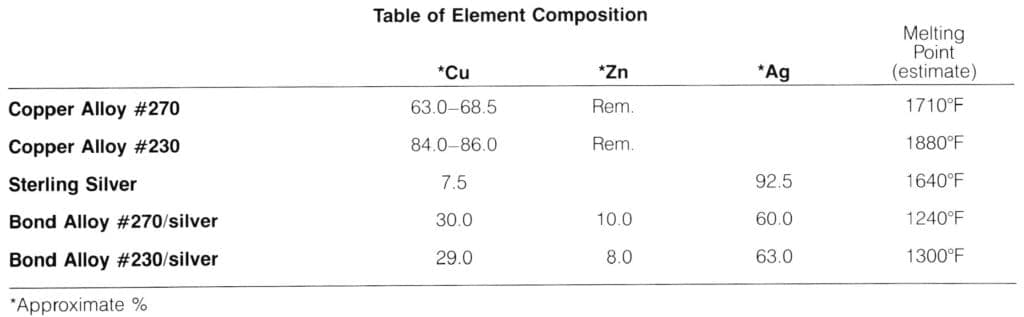
Fusion Patterns
12 Minute Read
This paper describes a process that combines techniques researched by others with a few well known facts about the properties of certain metals. This information was applied to a metal fabrication process specifically designed to extend the images and forms that interest me.
In presenting this paper it is my hope that others will find applications appropriate to their own work. I make this point because I know that we are already aware of quantities of techniques and processes, and I feel reticent about describing yet another when I see a much stronger need for development of ideas to apply them all to.
Fusion Patterns
My work, for the last few years, has been concerned with literal images that include elements of pattern and fabric-like form. I have explored many different media with variations of these elements as a constant—fabric and metal, fiberglass constructions and casts, cement and, most recently, life size figurative repoussé die forms painted with automotive lacquers. The photoetching and fusion processes provided an ideal method for producing a manipulable, patterned sheet stock for use in jewelry and small scale pieces.

Plating, married metals, soldered laminations and mokume-gane can all be used to produce multicolored sheet metal. However, each was inappropriate for my purposes. I wanted complex, controlled patterning that could be rolled, twisted, repousséd, stretched and raised. The phenomenon of brass/silver fusion at unexpectedly low temperatures is usually regarded as a problem. To me, instead, it seemed to offer a solution to producing the kind of multicolored sheets I required.
This bonding of brass and silver can be defined as fusion welding, in that the bond is formed using heat but not solder. It occurs when these metals are brought to a molten state at their contacting surfaces. Oppi Untracht defines the term in his book Jewelry Concepts and Technology (page 348) as follows. "The term welding implies that the surfaces are joined in a molten state, and fusion implies that an interpenetration of atoms occurs."
Background
Experimentation began with "found" perforated (approximately .0025″) brass sheet. Variously patterned sheets of this very thin etched brass turned up at the scrapyard, discarded by local electronics manufacturers. Several attempts were made to unite different gauges of sterling silver with these industrially produced patterns. These early experiments produced varying results. In general, the thin stock was an unsatisfactory material. The welding process was very touchy. The etched piece moved a great deal, and patience, skill and pure luck were required to make it fuse to a thicker sheet of silver without melting. The brass sheet often sank suddenly and unexpectedly into the silver surface in a yellowish mass. Also, rolling, repoussé and other forming processes were severely limited due to the thinness of the patterned brass layer.

To reproduce patterns of my own choice, I theorized that a thicker brass stock would be not only more stable but less likely to fuse suddenly and completely. Experimentation with different patterns, alloys and metal gauges was necessary. My experiments showed that thicker brass sheets would indeed be more easily fused to the silver. However, as the thickness of the brass sheet increased, another problem developed. The etched perforations were smaller on the back surface of the piece than on the front. This resulted in unacceptable distortions of the pattern. Therefore, it was necessary to find the optimum brass thickness which would be thin enough to permit an even etch from front to back.
This problem can be eliminated by the use of exposures with the same transparency on the front and back of a plate that has been completely coated with emulsion. The etch then proceeds from both surfaces toward the middle. In order to use this method, the exposures have to be made with the transparency in perfect register. Otherwise the etched perforations would be misaligned from front to back. (The process and an easily constructed, portable spray etcher were presented by Linda Threadgill at the 1983 Metals Symposium in Yuma, Arizona. See Suppliers.)
The Process
Etching
The transparency must have a completely "connected" pattern. For example, an etched "O" would become a round hole when the circular disk of metal dropped out of the center. Inattention to this problem can result in a large collection of unusable little scraps of metal and a greatly simplified pattern.
The brass plate can be coated, exposed, developed and etched using normal metal photoetching processes. Ammonium persulphate etching mordant is particularly suited to this technique, as it provides a clear, precise image that varies little from "front" to "back" of the plate. It is important to cover the emulsion-free "back" of the plate with transparent tape or contact paper that will allow clear observation of the progress of the etch through the plate. (Special tape for this purpose can be obtained from Bill Seeley, Reactive Metals Studio, see Suppliers.) When the pattern is etched through, creating a satisfactory pierced image, the plate should be cleaned thoroughly with lacquer thinner, annealed, pickled and pumiced. This task must be accomplished with great care in supporting what is now a fragile screen of thin brass.
Fusing
A piece of silver o{ appropriate gauge (16 - 22 are easiest to work with), is cut to match the brass plate, Both brass and silver should be absolutely flat and clean. The etched brass plate is laid on the silver. Silver and brass are fluxed thoroughly. The sandwiched metal is heated slowly and evenly. If the brass begins to lift, it should be pressed back gently into contact with the silver, using the side of a solder pick. Where silver and brass meet, the edges will shimmer or flush like a solder join. Immediately after this fusion takes place, the brass will begin to sink into the silver surface. To avoid this, remove the flame immediately and move on to unfused areas until the whole piece is without bubbles or open edges.
Pickle and clean as usual. Overheated areas will have sunk in and become flooded with the new alloy formed in the fusion zone. Some such areas can be sanded to restore the pattern lf the new alloy has only flowed over the brass without disturbing its form. If air bubbles are visible, the metal can be pierced, fluxed and reheated until it fuses.
Experimentation
For the sake of consistency I have adopted the Carbondale vocabulary from "Introduction to Granulation," SIU, Carbondale (see Bibliography), to describe bonding characteristics in these experiments. These characteristics are: good readability at the fusion point, clean surfaces without reticulation or pitting and acceptable bond strength with small fillets.
The brass was etched with ferric chloride or ammonium persulphate. A natural gas/oxygen torch was the preferred method for heating in all cases, as it afforded a flexible and immediately observable means of controlling the process. Twenty-gauge (.032″) silver was the base plate in all test pieces. Copper alloy #270 (yellow brass) and copper alloy #230 (red brass) were chosen for experimentation because of their qualities of workability and easy availability to the craftsman. Very flat yellow brass with an immaculate surface in suitable gauges is available from K&S Engineering (see Suppliers). They also supply the same material to most model and hobby shops.
Copper Alloy #270
A .005″ — Etch: Even and clean through the sheet. Fusion: Some movement and warping could be controlled with even heating and limited Pressure with a solder pic. Good readability, clean surfaces, some variation in fillet sizes.
B .015″ — Etch: Slightly uneven, with somewhat smaller image on back of sheet. Fusion: Etched sheet more stable when heated, good readability, clean surfaces and little variation in fillet size.
C .032″ — Etch: Image on back of sheet unacceptably smaller with loss of detail. Fusion: Fusion with a substituted piece of manually pierced metal was very easy to control, exhibited good readability, clean surfaces and little variation in fillet size.
Copper Alloy #230
A .005″ — Etch: Even and clean through the sheet. Fusion: Some movement and warping, good readability, clean surfaces and less variation in fillet size than a brass of same gauge.
B .015″ — Etch: Slightly uneven, with somewhat smaller image on back of sheet. Fusion: Etched sheet more stable when heated, good readability, clean surfaces and little variation in fillet size.
C .032″ — Etch: Image on back of sheet unacceptably smaller with loss of detail. Fusion: Fusion with a substituted piece of manually pierced metal was very easy to control, exhibited good readability, clean surfaces and little variation in fillet size.
Advantages and Disadvantages
Pieces of this fused material have been rolled, twisted, repoussed and chased with good results. The brass surface can be heat treated and otherwise patinaed to a wide range of colors. Due to the nature of the alloy produced at the interface of the two metals, one is limited to the use of low-temperature solder or cold connections in further construction. However, this limitation has had little influence on complexity or lack of it in pieces made with fused material.

Composition of the bond alloy is radically different from the laminates and may result in some weakness in certain structures. This has not been a problem in this experimentation, however.
Some very small spaces in the pattern flood with the bonding alloy which has a slightly different texture and color than the rest of the sterling silver. Rolling unifies the texture, so color difference is hardly detectable.
There is a definite size limitation with the torch fusing method. Laminations larger than four-inches square become very unwieldy due to movement of the perforated sheet caused by uneven heating.
There is limit to the "stretch" of the pattern that is determined by the thickness of the perforated sheets. The double-sided etching process produces cleanly perforated sheets and this makes it possible to etch thicker sheets, which extends workability. However, pattern distortion will increase as the material is "stretched" further. Very fine patterns will still be limited to thinner gauges, even with a two-sided etch Patterns that incorporate perforations of radically different size present problems in etchinq. Smaller spaces generally take longer to etch; meanwhile the larger areas will spread and lose detail. Selective "stop, ping out" with lacquer, nail polish or asphaltum during etching will eliminate this problem
Conclusions
The fusion-weld bonding of brass and silver does not quite fit the description of the process used in either mokume-gane or granulation. However, the results of energy dispersive x-ray analysis lead me to believe the phenomenon is more closely related to granulation than the heat/pressure induced results of mokume-gane. The existence of copper in both sterling silver and brass contributes to activity quite like the interpenetration of copper in the joining alloy of gold and silver granulation. The presence of zinc in the group probably lowers even further the temperature at which diffusion occurs. X-ray images of the fusion areas primarily show penetration of the silver layer by diffused atoms of copper. The edge of the silver layer on the other hand, remains relatively well-defined. Copper and zinc appear to be the active elements.
X ray analysis also provides evidence that suggests the smaller amount of zinc in copper alloy #230 may be the reason for a more controllable fusion of that alloy with sterling silver. The new alloy forms between the two layers at a somewhat higher temperature and does not flow as easily or quickly. Working qualities of #230 are closer to those of sterling silver and so contribute to a more unified metal body.
Further experimentation will certainly yield more precision and variation in this process. Other metals (most notably alloys of gold and silver) with similar attributes present new avenues for technical exploration. Even more exciting to me are the possibilities this process suggests in terms of imagery and surface enrichment. They are as limitless as the number of artists interested in applying them to their own visual development.
Bibliography
- Avner, Sidney H., Introduction to Physical Metallurgy, New York: McGraw-Hill, 1964.
- Copper Development Association, Inc., Standards Handbook, Wrought Mill Products, Alloy Datal2, New York, 1973.
- Eastman Kodak Company, Kodak Master Photoguide, publication AR-21, New York, March, 1981.
- Evans, Chuck, Jewelry, Contemporary Design and Technique, Worcester, MA: Davis Publications, 1983.
- Members of the Graduate Program, Southern Illinois University — Carbondale; compiled and edited by W. Ard, "Return to the Forge (extended research into mokume-gane and granulation)," Goldsmith's Journal, August, 1979; reprinted in Metalsmith Papers of the Society of North American Goldsmiths, 1981.
- McCloskey, John C., "A Metallurgical Analysis of the Lamination of Non-Ferrous Metals," Goldsmith's Journal , February, 1978.
- Moty, Eleanor, "Workshop: Photofabrication," Craft Horizons, June 1971, Vol.31, No. 3, pp. 12-17
- Threadgill, Linda, "Alternative Etching Methods," Metalsmith, Fall, 1981, Vol. One, No. Four, p. 350.
- Untracht, Oppi, Jewelry, Concepts and Technology, New York: Doubleday, 1982.
- Wade, Kent, Alternative Photographic Processes, New York: Morgan and Morgan, 1978.
- Woell, J. Fred, Photography in the Crafts, J. Fred Woell, 14A Dow Road, Deer lsle, ME 04627, 1975.
Suppliers
- Brass — K&S Engineering, 6917 West 59th St., Chicago, IL 60638
- Photo Chemicals and Data Sheets — Thiokol/Dynachem, 3311 Boyinton Drive, Suite 380, Caroollton, TX 75006 Spray-etcher — Linda Threadgill, University of Wisconsin, Whitewater, WI 53190
- Light and Drying Boxes — Eleanor Moty, "Photofabrication on Metal," from Photography in the Crafts, J. Fred Woell
- Etching Tape — Reactive Metals Studio, P.O. Box 2591, Sedona, AZ 86336-M
Acknowledgments
Thanks to David Pimentel , Eleanor Moty and Richard Helzer for advice, encouragement and a great deal of useful information; to Dr. Joseph LaPointe of the NMSU Biology Department; and especially to Henry Adams of the NMSU Biology Department for his patience and skill in the application of electron microscopy to this project.
This project was funded initially by a research assistantship from Arizona State University. A generous grant from the Arts and Sciences Research Center at New Mexico State University has brought it to completion.
Kate Wagle is a metalsmith and Assistant Professor of Jewelry and Metalsmithing at New Mexico State University in Las Cruces, NM.

The All-In-One Jewelry Making Solution At Your Fingertips
When you join the Ganoksin community, you get the tools you need to take your work to the next level.